TESTING KITS
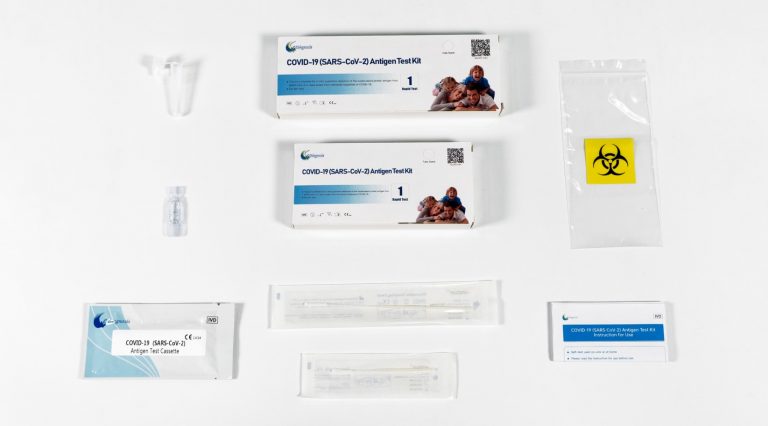
COVID-19(SARS-CoV-2) Antigen Test Kit
Reagent Parameters

PRODUCT #AZ-MED-T411 TEST KIT
Information:
- Result in 15 minutes
- Identifies acute or early infection
- CE certified
Performance Characteristic
- Clinical Sensitivity: 96.13%, 95% confidence interval: [92.05%, 97.98%]
- Clinical Specificity: 99.06%, 95% confidence interval: [97.72%, 99.53%]
- Clinical Accuracy: 98.28%, 95% confidence interval: [96.92%, 99.01%]
Main Components:
- Antigen Test Cassette
- Antigen Extract R1
- Anterior Nasal Swab
- Antigen Extraction Tube
- Tube Stand
- Biohazard Waste Bag
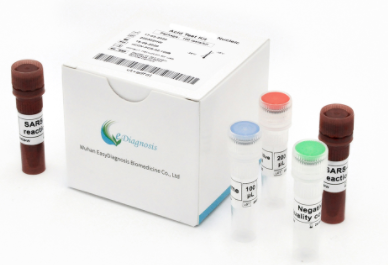
COVID – 19 Nucleic Acid Test Kit
PRODUCT #AZ-MED-T409 TEST KIT
- Fast: Optimized Reaction System, Greatly Shortening PCR Amplification Time, Result is available within 75minutes
- Good Performance: Clinical Accuracy 95.03%
- Reliable: ORF1ab gene and N gene dual-site detection
- Product name: COVID-19 (SARS-CoV-2) Nucleic Acid Test Kit
- Intended use: COVID-19 SARS-CoV-2 Nucleic Acid Test Kit is a real-time RT-PCR test kit, intended for the qualitative detection of nucleic acid from the SARS-CoV-2, in upper and lower respiratory tract specimens (such as oropharyngeal swabs, nasopharyngeal swabs and sputum).
- Storage conditions and Validity: 6 months at -20±5℃
- Instruments applied: ABI7500 (Thermo Fisher), LightCycler480 (Roche) and other quantitative fluorescence PCR platforms with FAM, HEX and ROX fluorescence detection channel.
- Package: 100 tests/kit
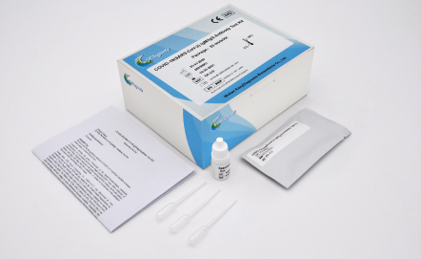
COVID-19 (SARS-CoV-2) IgG/IgM Antibody Test Kit
PRODUCT #AZ-MED-T410 TEST KIT
Information:
- Result in 15 minutes
- CE certified
Performance Characteristic
- Identify acute or early infection
- Sensitivity: 98.33%
- Specificity: 98.13%
- Accuracy: 98.18%
- Storage conditions and Validity:12months at 4℃~30℃
Package: 20 tests/kit and 40 tests/kit
COVID-19(SARS-CoV-2) Antigen Test Kit (Cont)
COVID-19 Antigen Test Procedure
1.Twist off the cap of the buffer bottle, carefully dispense all buffer into the extraction tube。
2.After collecting upper respiratory sample with nasal swab, insert the swab into the extraction tube, plunge the swab up and down in the fluid for a minimum of 10 seconds. Hold the swab against the bottom of the tube, rotate three turns. DO NOT splash liquid out of the tube.
3.Remove the swab while squeezing the sides of the tube to extract the liquid from the swab.
4.Press the nozzle cap firmly onto the extraction tube. Mix thoroughly by swirling or flicking the bottom of the tube.
5.Gently squeeze the tube’s rigid body, dispense two (2) drops of the buffer-specimen mixture into the sample well on the coronavirus antigen test cassette.
6.Read the test results between 15 and 20 minutes.
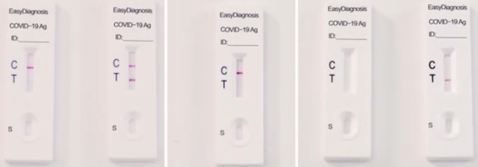
Interpretation of Test Results
1.NEGATIVE: A coloured band appears on the control line (C line); no coloured band shows up on the test line (T line). A negative result indicates there is no coronavirus antigen (N protein) in the specimen, or the level of coronavirus antigen is below the detection limit.
2.POSITIVE: A coloured band appears on the control line (C line), a second coloured band shows up on the test line (T line). A positive result indicates the presence of COVID-19 antigen (N protein) in the patient sample.
3.INVALID: No coloured band appears on the control line (C line). An invalid test result suggests there might be insufficient buffer volume or incorrect operating procedures. Carefully review the test procedure and test the same patient again with another coronavirus antigen rapid test cassette. Contact your distributor if the problem persists.
COVID-19 & Influenza A/B Nucleic Acid Test Kit
PRODUCT #AZ-MED-T412 TEST KIT
Information:
- Simultaneous detection of mulit-pathogens
- Early diagnosis of unknown fever
- Prevent cross infection
- CE certified
- Result in 90 minutes.
- Multi-pathogens detection: The influenza A/B virus and SARS-CoV-2 N gene are detected simultaneously in a single tube
- Reliable: Co-efficient of variation of Ct value CV<=5%, Simultaneous detection of human internal standard and negative-positive quality control products, monitor false positive and false negative events effectively.
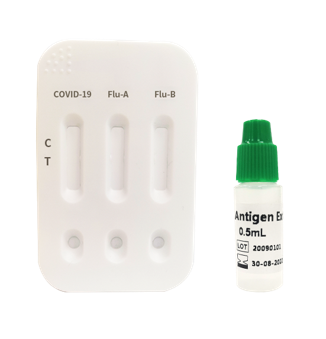
COVID-19 & Influenza A/B Antigen Combo Rapid Test Kit
PRODUCT #AZ-MED-T413 TEST KIT
Information:
- Result in 15 minutes.
- High sensitivity and specificity
Package: 1 test/kit and 20 test/kit

QPT1000 iPOCT Molecular Diagnosis System
PRODUCT #AZ-MED-T414 TEST KIT
Information:
- External dimensions: 398mm x 255mm x 272mm
- Weight: <10kg
- Touch Display Screen: 10.1-inch TFT, Capacitive Touch
- Operating System: Linux System
- Detection Channel: 2/4/8-Channel(optional), Each Channel Can Realize Independent Detection
Product Brief:
Directed at molecular target, this product adopts special temperature sequence for chain amplification reaction, and the amplification target molecule is labelled with luminous enzyme. After that, the luminescence signal is collected to determine the number of amplified molecular targets, and the number of molecular targets before amplification is deduced by intelligent algorithm Supports molecular level detection of virus, including COVID-2019
Application Scenarios: Hospitals, outdoors and field operations.
Product Features:
Sensitivity: Advanced optical inspection system to realize high-sensitivity fluorescence detection Accuracy: High-efficiency and stable temperature control system to realize accurate and rapid control of temperature rise and drop
Rapidity: Independent module control mode to realize differential amplification program and improve the detection efficiency
Intelligence: Advanced software system to realize intelligent operation and result analysis

QFT9000 Fluorescence Immunity Analyzer
PRODUCT #AZ-MED-T415 TEST KIT
Information:
- Result in 8 minutes
- Multi-sample-type Compatibility
- Thermostatic Reaction
- CE certified
Product Brief:
- QFT9000 Fluorescence Immunity Analyzer is used for interpretation of the dry type fluorescence immunoassay kit produced by our company, as well as clinical quantitative testing of analyte in whole blood, serum and plasma samples from human body.
- Whole Blood, Serum and Plasma.
- Reagents store at room temperature.

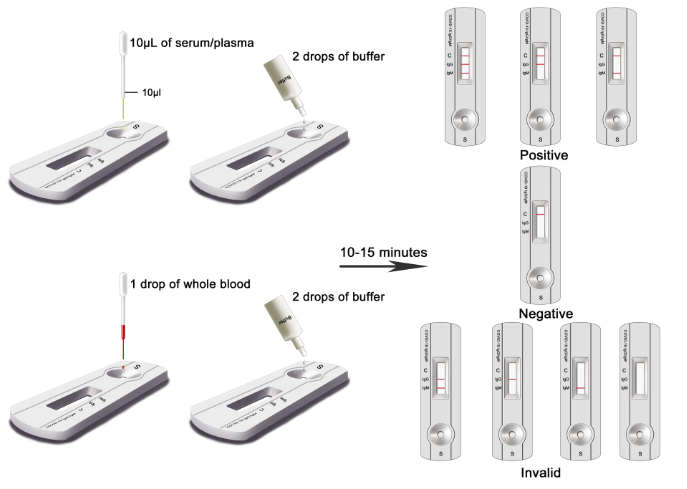
SARS-CoV-2 IgM/IgG Antibody Test Kit
- SARS-CoV2 Fast Detection Kit – Antibody Test Kit
PRODUCT #AZ-MED-120 TEST KIT
NeoPlex SARS-CoV-2 Detection Kit – (RT-PCR)
- SARS-CoV2 Fast Detection Kit
- Please enquire for user information
PRODUCT #AZ-MED-118 TEST KIT
KHB SARS-CoV-2 IgM/IgG Antibody Test Kit
- KHB SARS-COV-2 IGM/IGG Antibody test kit can be used for rapid screening of carriers of the virus that are symptomatic or asymptomatic.
- CE Certified
PRODUCT #AZ-MED-145 TEST KIT
Azure Aero Limited, is supporting the international efforts to supply critical Medical PPE to the front line of medical staff wherever they may be. Please contact us through our contact form below or on our contact number +44-7624-329115 to purchase products and for further information on items and availability. We will do our best to support you.
AZURE AERO MEDICAL SUPPORT ORDER & ENQUIRY FORM
Please fill out this form to enquire about our products and to receive an order quotation. We will be in contact about your request.



